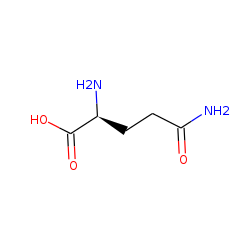
L-Glutamine CAS:56-89-5 98.5%~101.0%
|
Cas.No |
56-85-9 |
|
Assay |
99.0-101.0% |
|
Description |
White crystals or crystalline powder, odorless, slightly characteristic taste |
|
Uses |
Pharmaceutical, Cell culture media |
|
Packaging |
25kg&50kg |
|
Pharmacopeia |
JP,USP,FCC |
|
STORAGE |
Controlled room temperature in tight container |
SPECIFICATIONAND PROCEDURE
State of solution
(Transmittance) |
Not Less Than 98.0% |
|
pH |
4.0~6.0 |
|
Specific rotation[α]20D |
+6.3~+7.3° |
|
Chloride (Cl) |
Not More Than 0.020% |
|
Sulfate (SO4) |
Not More Than 0.020% |
|
Iron (Fe) |
Not More Than 10 ppm |
|
Heavy metals (Pb)** |
Not More Than 10 ppm |
|
Arsenic (As2O3) |
Not More Than 1 ppm |
|
Loss on drying |
Not More Than 0.20% |
|
Residue on ignition |
Not More Than 0.10% |
|
Related substances |
Not More Than 0.5% |
|
Endotoxin* |
Less Than 6.0 EU/g |
|
Assay (dry basis) |
99.0~101.0% |
A non-essential amino acid present abundantly throughout the body and is involved in many metabolic processes. It is synthesized from glutamic acid and ammonia. It is the principal carrier of nitrogen in the body and is an important energy source for many cells. An oral formulation of L-glutamine was approved by the FDA in July 2017 for use in sickle cell disease 5. This oral formulation is marketed under the tradename Endari by Emmaus Medical.
If you are looking for L-Glutamine CAS:56-89-5 producer, Glutamic acid amide manufacturer,Glutamic acid 5-amide factory or L-(+)-glutamine bulk supplier, and get (2S)-2,5-diamino-5-oxopentanoic acid price information, please contact us to get L-Glutamine COA (Certificate of Analysis,MSDS(Material safety data), we are also offering custom synthesis and contract manufacturing services.